Matching
|
|
|
Match each item with the correct statement below. a. | coordinate covalent bond | d. | single covalent
bond | b. | double covalent bond | e. | polar bond | c. | structural formula | f. | hydrogen bond |
|
|
|
1.
|
a depiction of the arrangement of atoms in molecules and polyatomic
ions
|
|
|
2.
|
a covalent bond in which only one pair of electrons is shared
|
|
|
3.
|
a covalent bond in which two pairs of electrons are shared
|
|
|
4.
|
a covalent bond in which the shared electron pair comes from only one of the
atoms
|
|
|
5.
|
a covalent bond between two atoms of significantly different
electronegativities
|
|
|
6.
|
a type of bond that is very important in determining the properties of water
and of important biological molecules such as proteins and DNA
|
Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
7.
|
Which is a typical characteristic of an ionic compound?
a. | Electron pairs are shared among atoms. | b. | The ionic compound has a low solubility in
water. | c. | The ionic compound is described as a molecule. | d. | The ionic compound
has a high melting point. |
|
|
|
8.
|
What is shown by the structural formula of a molecule or polyatomic ion?
a. | the arrangement of bonded atoms | c. | the number of metallic
bonds | b. | the number of ionic bonds | d. | the shapes of molecular orbitals |
|
|
|
9.
|
Which of these elements does not exist as a diatomic molecule?
|
|
|
10.
|
How do atoms achieve noble-gas electron configurations in single covalent
bonds?
a. | One atom completely loses two electrons to the other atom in the
bond. | b. | Two atoms share two pairs of electrons. | c. | Two atoms share two
electrons. | d. | Two atoms share one electron. |
|
|
|
11.
|
Why do atoms share electrons in covalent bonds?
a. | to become ions and attract each other | b. | to attain a noble-gas electron
configuration | c. | to become more polar | d. | to increase their atomic
numbers |
|
|
|
12.
|
Which of the following elements can form diatomic molecules held together by
triple covalent bonds?
a. | carbon | c. | fluorine | b. | oxygen | d. | nitrogen |
|
|
|
13.
|
Which noble gas has the same electron configuration as the oxygen in a water
molecule?
a. | helium | c. | argon | b. | neon | d. | xenon |
|
|
|
14.
|
Which elements can form diatomic molecules joined by a single covalent
bond?
a. | hydrogen only | b. | halogens only | c. | halogens and members
of the oxygen group only | d. | hydrogen and the halogens
only |
|
|
|
15.
|
Which of the following is the name given to the pairs of valence electrons that
do not participate in bonding in diatomic oxygen molecules?
a. | unvalenced pair | c. | inner pair | b. | outer pair | d. | unshared pair |
|
|
|
16.
|
Which of the following electron configurations gives the correct arrangement of
the four valence electrons of the carbon atom in the molecule methane (CH 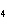 )?
|
|
|
17.
|
Which of the following diatomic molecules is joined by a double covalent
bond?
|
|
|
18.
|
A molecule with a single covalent bond is ____.
|
|
|
19.
|
When one atom contributes both bonding electrons in a single covalent bond, the
bond is called a(n) ____.
a. | one-sided covalent bond | c. | coordinate covalent
bond | b. | unequal covalent bond | d. | ionic covalent bond |
|
|
|
20.
|
Once formed, how are coordinate covalent bonds different from other covalent
bonds?
a. | They are stronger. | c. | They are weaker. | b. | They are more ionic in
character. | d. | There is no
difference. |
|
|
|
21.
|
When H 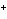 forms a bond with H 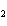 O to
form the hydronium ion H 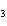 O  , this bond is called a
coordinate covalent bond because ____. a. | both bonding electrons come from the oxygen atom | b. | it forms an
especially strong bond | c. | the electrons are equally
shared | d. | the oxygen no longer has eight valence electrons |
|
|
|
22.
|
Which of the following bonds is the least reactive?
|
|
|
23.
|
How many valid electron dot formulas—having the same number of electron
pairs for a molecule or ion—can be written when a resonance structure occurs?
a. | 0 | c. | 2 only | b. | 1 only | d. | 2 or more |
|
|
|
24.
|
In which of the following compounds is the octet expanded to include 12
electrons?
|
|
|
25.
|
How many electrons can occupy a single molecular orbital?
|
|
|
26.
|
Molecular orbital theory is based upon which of the following models of the
atom?
a. | classical mechanical model | c. | quantum mechanical
model | b. | Bohr model | d. | Democritus’s model |
|
|
|
27.
|
A bond that is not symmetrical along the axis between two atomic nuclei is a(n)
____.
a. | alpha bond | c. | pi bond | b. | sigma bond | d. | beta bond |
|
|
|
28.
|
How is a pair of molecular orbitals formed?
a. | by the splitting of a single atomic orbital | b. | by the reproduction
of a single atomic orbital | c. | by the overlap of two atomic orbitals from the
same atom | d. | by the overlap of two atomic orbitals from different
atoms |
|
|
|
29.
|
The side-by-side overlap of p orbitals produces what kind of bond?
a. | alpha bond | c. | pi bond | b. | beta bond | d. | sigma bond |
|
|
|
30.
|
Where are the electrons most probably located in a molecular bonding
orbital?
a. | anywhere in the orbital | b. | between the two atomic
nuclei | c. | in stationary positions between the two atomic nuclei | d. | in circular orbits
around each nucleus |
|