Matching
|
|
|
Match each item with the correct statement below. a. | mass number | d. | atomic mass | b. | atomic mass unit | e. | isotope | c. | atomic
number |
|
|
|
1.
|
atoms with the same number of protons, but different numbers of neutrons in the
nucleus of an atom
|
|
|
2.
|
the total number of protons and neutrons in the nucleus of an atom
|
|
|
3.
|
the number of protons in the nucleus of an element
|
|
|
4.
|
the weighted average of the masses of the isotopes of an element
|
|
|
5.
|
one-twelfth the mass of a carbon atom having six protons and six
neutrons
|
Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
6.
|
An element has an atomic number of 76. The number of protons and electrons in a
neutral atom of the element are ____.
a. | 152 protons and 76 electrons | c. | 38 protons and 38
electrons | b. | 76 protons and 0 electrons | d. | 76 protons and 76 electrons |
|
|
|
7.
|
The sum of the protons and neutrons in an atom equals the ____.
a. | atomic number | c. | atomic mass | b. | nucleus number | d. | mass number |
|
|
|
8.
|
What does the number 84 in the name krypton-84 represent?
a. | the atomic number | c. | the sum of the protons and electrons | b. | the mass
number | d. | twice the number of
protons |
|
|
|
9.
|
All atoms of the same element have the same ____.
a. | number of neutrons | c. | mass numbers | b. | number of protons | d. | mass |
|
|
|
10.
|
Isotopes of the same element have different ____.
a. | numbers of neutrons | c. | numbers of electrons | b. | numbers of protons | d. | atomic numbers |
|
|
|
11.
|
Isotopes of the same element have different ____.
a. | positions on the periodic table | c. | atomic numbers | b. | chemical
behavior | d. | mass
numbers |
|
|
|
12.
|
In which of the following sets is the symbol of the element, the number of
protons, and the number of electrons given correctly?
a. | In, 49 protons, 49 electrons | c. | Cs, 55 protons, 132.9
electrons | b. | Zn, 30 protons, 60 electrons | d. | F, 19 protons, 19
electrons |
|
|
|
13.
|
The mass number of an element is equal to ____.
a. | the total number of electrons in the nucleus | b. | the total number of
protons and neutrons in the nucleus | c. | less than twice the atomic
number | d. | a constant number for the lighter elements |
|
|
|
14.
|
Using the periodic table, determine the number of neutrons in 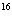 O.
|
|
|
15.
|
How many protons, electrons, and neutrons does an atom with atomic number 50 and
mass number 125 contain?
a. | 50 protons, 50 electrons, 75 neutrons | c. | 120 neutrons, 50 protons, 75
electrons | b. | 75 electrons, 50 protons, 50 neutrons | d. | 70 neutrons, 75 protons, 50
electrons |
|
|
|
16.
|
Which of the following statements is NOT true?
a. | Atoms of the same element can have different masses. | b. | Atoms of isotopes of
an element have different numbers of protons. | c. | The nucleus of an atom has a positive
charge. | d. | Atoms are mostly empty space. |
|
|
|
17.
|
If E is the symbol for an element, which two of the following symbols represent
isotopes of the same element? a. | 1 and 2 | c. | 1 and 4 | b. | 3 and 4 | d. | 2 and 3 |
|
|
|
18.
|
Select the correct symbol for an atom of tritium.
|
|
|
19.
|
Which of the following sets of symbols represents isotopes of the same
element?
|
|
|
20.
|
How is the number of neutrons in the nucleus of an atom calculated?
a. | Add the number of electrons and protons together. | b. | Subtract the number
of electrons from the number of protons. | c. | Subtract the number of protons from the mass
number. | d. | Add the mass number to the number of electrons. |
|
|
|
21.
|
In which of the following is the number of neutrons correctly
represented?
a. | 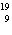 F has 0 neutrons. F has 0 neutrons. | c. | 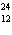 Mg has 24
neutrons. Mg has 24
neutrons. | b. | 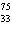 As has 108 neutrons. As has 108 neutrons. | d. | 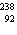 U has 146
neutrons. U has 146
neutrons. |
|
|
|
22.
|
How do the isotopes hydrogen-1 and hydrogen-2 differ?
a. | Hydrogen-2 has one more electron than hydrogen-1. | b. | Hydrogen-2 has one
neutron; hydrogen-1 has none. | c. | Hydrogen-2 has two protons; hydrogen-1 has
one. | d. | Hydrogen-2 has one proton; hydrogen-1 has none. |
|
|
|
23.
|
Which of the following isotopes has the same number of neutrons as
phosphorus-31?
|
|
|
24.
|
What unit is used to measure weighted average atomic mass?
a. | amu | c. | angstrom | b. | gram | d. | nanogram |
|
|
|
25.
|
Which of the following equals one atomic mass unit?
a. | the mass of one electron | b. | the mass of one helium-4
atom | c. | the mass of one carbon-12 atom | d. | one-twelfth the mass of one carbon-12
atom |
|
Numeric Response
|
|
|
26.
|
How many protons are present in an atom of Be-9?
|
|
|
27.
|
What is the total number of subatomic particles in the nucleus of an atom of
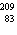 Bi?
|
|
|
28.
|
Determine the number of electrons in an atom of iridium.
|
|
|
29.
|
What is the atomic number for an element with 41 neutrons and a mass number of
80?
|
|
|
30.
|
How many electrons are in an atom of gold?
|