Matching
|
|
|
Match each item with the correct statement below. a. | product | d. | balanced equation | b. | reactant | e. | skeleton equation | c. | chemical
equation |
|
|
|
1.
|
a chemical equation that does not indicate relative amounts of reactants and
products
|
|
|
2.
|
a new substance formed in a chemical reaction
|
|
|
3.
|
a starting substance in a chemical reaction
|
|
|
4.
|
a concise representation of a chemical reaction
|
|
|
5.
|
an equation in which each side has the same number of atoms of each
element
|
|
|
Match each item with the correct statement below. a. | activity series of metals | c. | combustion
reaction | b. | single-replacement reaction | d. | decomposition reaction |
|
|
|
6.
|
a reaction in which a single compound is broken down into simpler
substances
|
|
|
7.
|
a reaction in which oxygen reacts with another substance, often producing heat
or light
|
|
|
8.
|
a reaction in which the atoms of one element replace the atoms of a second
element in a compound
|
|
|
9.
|
a list of metals in order of decreasing reactivity
|
Multiple Choice
Identify the
choice that best completes the statement or answers the question.
|
|
|
10.
|
Chemical reactions ____.
a. | occur only in living organisms | c. | only occur outside living
organisms | b. | create and destroy atoms | d. | produce new substances |
|
|
|
11.
|
What does the symbol  in a chemical
equation mean? a. | Heat is supplied to the reaction. | c. | yields | b. | A catalyst is
needed. | d. | precipitate |
|
|
|
12.
|
A skeleton equation does NOT show which of the following?
a. | the correct formulas of the reactants and products | b. | the reactants on the
left, the products on the right | c. | an arrow connecting the reactants to the
products | d. | the relative amounts of reactants and products |
|
|
|
13.
|
Symbols used in equations, together with the explanations of the symbols, are
shown below. Which set is correct?
a. | (g), grams | c. | (aq), dissolved in water | b. | (l),
liters | d. | (s), solid
product |
|
|
|
14.
|
In the chemical equation H 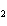 O 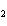 ( aq) ® H 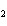 O( l)
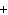 O 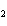 ( g), the 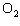 is a ____. a. | catalyst | c. | product | b. | solid | d. | reactant |
|
|
|
15.
|
This symbol (  ) indicates that ____. a. | heat must be applied | b. | an incomplete combustion reaction has
occurred | c. | a gas is formed by the reaction | d. | the reaction is
reversible |
|
|
|
16.
|
A catalyst is ____.
a. | the product of a combustion reaction | b. | not used up in a reaction | c. | one of the reactants
in single-replacement reactions | d. | a solid product of a
reaction |
|
|
|
17.
|
Which of the following is the correct skeleton equation for the reaction that
takes place when solid phosphorus combines with oxygen gas to form diphosphorus pentoxide?
|
|
|
18.
|
If you rewrite the following word equation as a balanced chemical equation, what
will the coefficient and symbol for fluorine be? nitrogen trifluoride 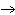 nitrogen 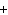 fluorine
|
|
|
19.
|
What are the coefficients that will balance the skeleton equation
below? AlCl 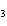 + NaOH 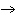
Al(OH) 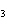 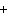 NaCl a. | 1, 3, 1, 3 | c. | 1, 1, 1, 3 | b. | 3, 1, 3, 1 | d. | 1, 3, 3, 1 |
|
|
|
20.
|
What are the coefficients that will balance the skeleton equation below?
N 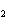 + H 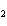 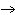 NH 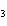 a. | 1, 1, 2 | c. | 3, 1, 2 | b. | 1, 3, 3 | d. | 1, 3, 2 |
|
|
|
21.
|
When the equation Fe 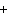 Cl 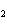 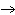 FeCl 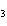 is balanced, what is the coefficient for
Cl 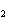 ?
|
|
|
22.
|
When the following equation is balanced, what is the coefficient for HCl?
Mg( s) 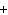 HCl( aq) 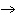
MgCl 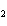 ( aq) 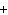 H 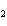 ( g)
|
|
|
23.
|
Which of the following statements is NOT true about what happens in all chemical
reactions?
a. | The ways in which atoms are joined together are changed. | b. | New atoms are formed
as products. | c. | The starting substances are called reactants. | d. | The bonds of the
reactants are broken and new bonds of the products are formed. |
|
|
|
24.
|
Chemical equations must be balanced to satisfy ____.
a. | the law of definite proportions | c. | the law of conservation of
mass | b. | the law of multiple proportions | d. | Avogadro’s principle
|
|
|
|
25.
|
When the equation KClO 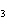 ( s) 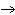 KCl( s) + O 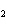 ( g) is balanced, the coefficient of KClO 3 is ____.
|
|
|
26.
|
In every balanced chemical equation, each side of the equation has the same
number of ____.
a. | atoms of each element | c. | moles | b. | molecules | d. | coefficients |
|
|
|
27.
|
What are the missing coefficients for the skeleton equation
below? Cr( s) 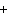 Fe(NO 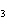 ) 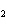 ( aq) 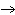 Fe( s) 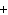
Cr(NO 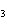 ) 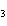 ( aq) a. | 4, 6, 6, 2 | c. | 2, 3, 3, 2 | b. | 2, 3, 2, 3 | d. | 1, 3, 3, 1 |
|
|
|
28.
|
What are the missing coefficients for the skeleton equation below?
Al 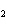 (SO 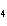 ) 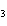 ( aq) 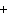 KOH( aq) ®
Al(OH) 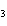 ( aq) 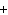 K 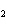 SO 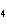 ( aq) a. | 1, 3, 2, 3 | c. | 4, 6, 2, 3 | b. | 2, 12, 4, 6 | d. | 1, 6, 2, 3 |
|
|
|
29.
|
When potassium hydroxide and barium chloride react, potassium chloride and
barium hydroxide are formed. The balanced equation for this reaction is ____.
|
|
|
30.
|
The product of a combination reaction is Ba(OH) 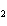 . If one of the
reactants is H 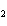 O, what is the other reactant? a. | Ba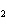 O O | c. | BaH | b. | BaO | d. | BaO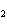 |
|